Identification of factors modifying disease severity and sudden cardiac death risk in inherited arrhythmia syndromes
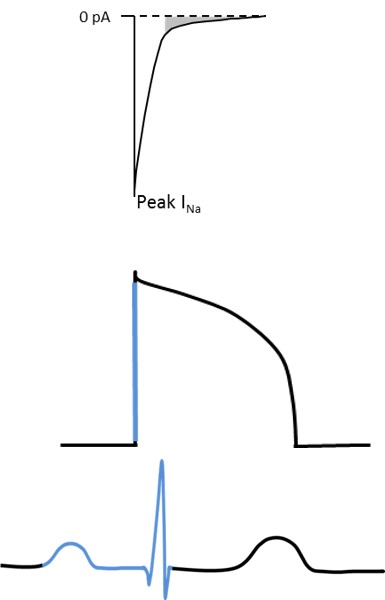
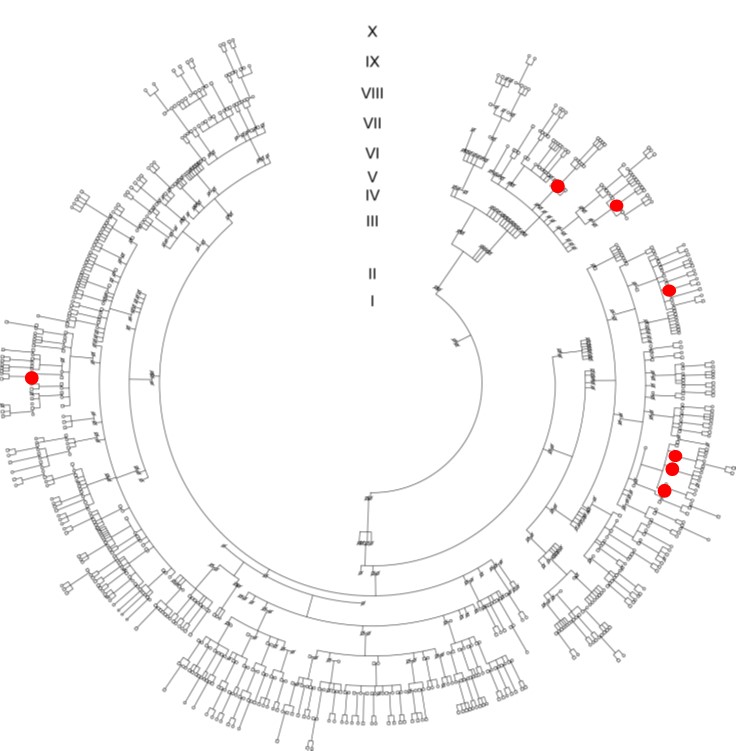
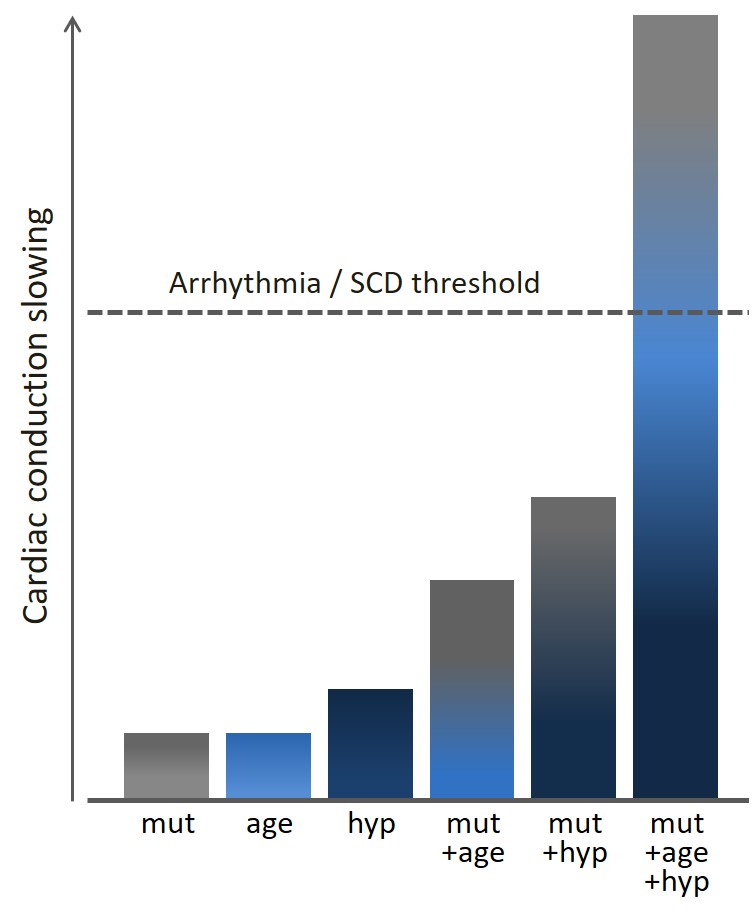
Significant variability in disease expression has been documented in patients harboring mutations in SCN5A, even among family members carrying the same mutation. Some mutation carriers suffer from arrhythmias and/or sudden death while others remain symptom-free throughout life. Gender and age have been shown to modulate disease severity, with age-related fibrosis thought to play a major role in exacerbating cardiac conduction with advancing age. In addition, a potential role for genetic modifiers has been suggested, including genetic variation derived from single nucleotide polymorphisms frequently observed in the general population. We have previously investigated the effects of genetic factors on disease severity by comparing the phenotype in two distinct strains of mice carrying the Scn5a-1798insD/+ mutation equivalent to SCN5A-1795insD in humans.
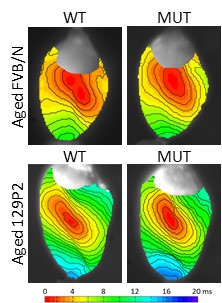
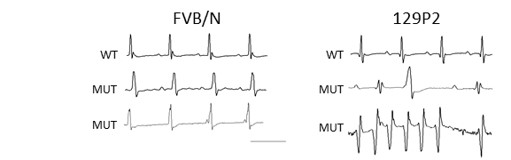
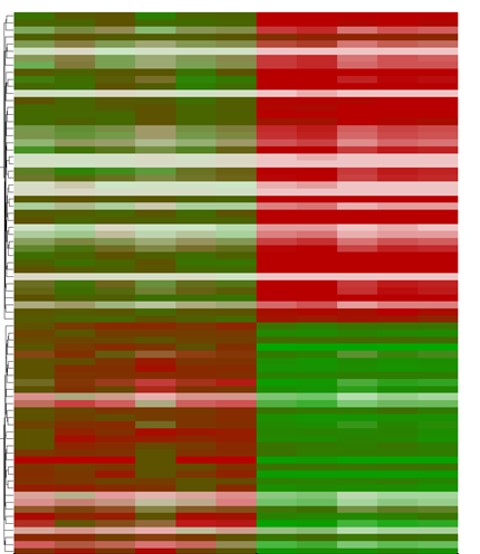
We found that phenotype severity was more pronounced in 129P2 mice as compared to FVBN/J mice and subsequently identified potential modifiers of conduction disease severity, providing the first conclusive evidence that genetic modifiers may determine disease expressivity in cardiac sodium channelopathy (Remme et al. Circ Res 2009). In a more recent study, we have also demonstrated that a common co-morbidity such as hypertension may modulate arrhythmia risk and treatment efficacy in inherited sodium channelopathy (Rivaud et al. Eur Heart J 2018). These observations emphasize the need for continued assessment and rigorous treatment of this co-morbidity in SCN5A mutation-positive individuals.
Our current research is aimed at identifying additional (genetic) factors and co-morbidities modulating arrhythmia and sudden cardiac death risk in patients with SCN5A mutations.
Group members
Carol Ann Remme
Group Leader, PI
Vincent Portero
Post Doc
Simona Casini
Post Doc
Giovanna Nasilli
PhD Student
Gerard Marchal
PhD Student